
What is n-Stride

n-STRIDE autologous protein solution (APS) is a novel minimally invasive, non-surgical treatment for joint pain and osteoarthritis (OA). APS has proven to be a safe and effective treatment and has been shown to reduce pain and protect cartilage by reducing further destruction.
This new approach to treating OA is based on our understanding of how knee OA progresses. In normal cartilage metabolism, there is a steady state between catabolic pro-inflammatory cytokines and anabolic growth factors.
In OA there is a degenerative “positive feedback loop” caused by pathological increases in Pro-inflammatory cytokines and catabolic factors (interleukin-1 beta (IL-1b), tumour necrosis factor-alpha (TNFa), and matrix metalloproteinases (MMPs).
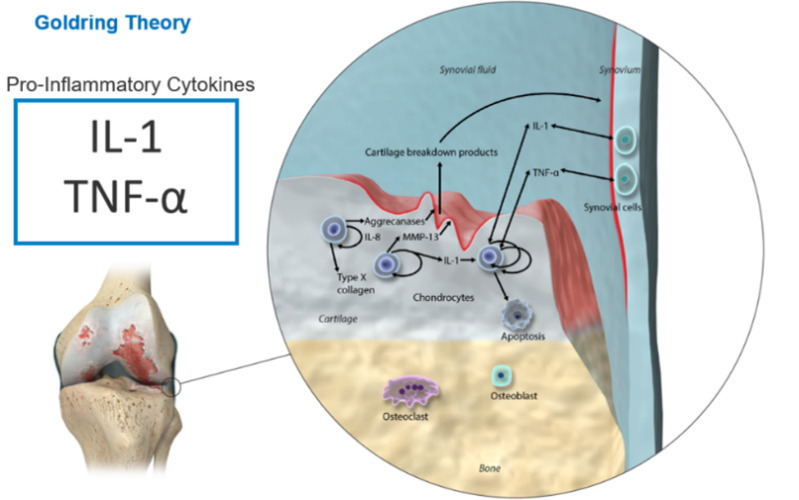
This causes an imbalance between the “steady-state” of cartilage remodelling, which leads to a disturbance of chondrocyte activity and loss of cartilage.
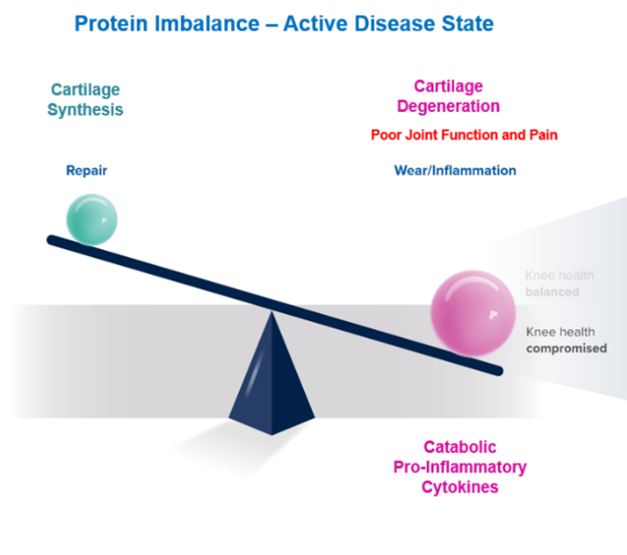
Approaches to block the deleterious action or production of inflammatory, catabolic proteins with APS, has been shown to improve patients’ symptoms, and perhaps the progression of the disease may be halted.
Indication for n-Stride APS Treatment
- Chronic knee pain secondary to osteoarthritis
- Poor response to conservative management
- Patients who are too young for knee replacement
- Patients who are unfit for knee replacement
- Patients who wish to delay or avoid surgery
How Do I Prepare for the Procedure?
- Avoid anti-inflammatories (ibuprofen, etc.) for two weeks before and two weeks after your injection.
- Do not stop taking low-dose aspirin if prescribed by your doctor.
- Take all prescribed medications as directed.
- This procedure is contraindicated for patients with inflammatory arthritis (i.e., rheumatoid arthritis).
- You are encouraged to have a small snack one hour before your procedure.
- If you take insulin to manage your diabetes, please let your booking coordinator know. You may have some juice and a small snack after taking your insulin.
- Wear comfortable clothing.
How is the Procedure Performed?
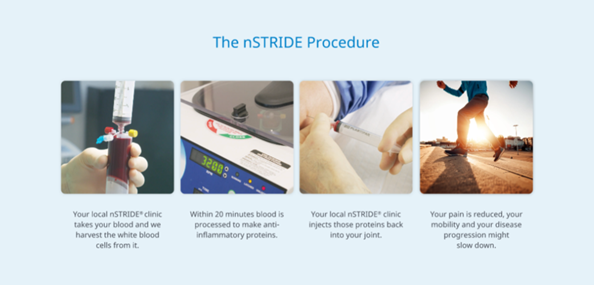
- The procedure is performed on an out-patient basis
- A Nurse will go over your consent form, explain the procedure, and answer any questions you may have.
- A small volume of blood will be withdrawn from your arm to prepare the injection.
- Using a centrifuge to separate the platelet-rich plasma in the blood, the plasma is then further processed with a centrifuge to separate a concentrated protein solution. You can rest in the waiting room during this process; it takes 10-20 minutes.
- A Nurse will bring you into the treatment room and position you on the table.
- Using image guidance, the Doctor will inject the APS preparation into the affected joint.
- You will be assessed post-injection by our team, and if there are no concerns, you will be free to leave.
Pain relief is usually experienced after around two weeks, but it is recommended that the knee is rested for a fortnight following the procedure.
Complications
The procedure is generally well-tolerated and the adverse effects reported in studies were generally mild. The side effects were largely related to the injection procedure itself. They included:
- Injection site discomfort
- Pain in the joint
- Joint stiffness
- Procedural nausea
These adverse effects tend to settle spontaneously and generally do not require any specific treatments.
When Will I See Results?
- Results from early healing may be appreciated after 1-2 weeks, although healing and repair will continue gradually for many months.
- Continued symptoms improvement is expected for up to 36 months. If an injection does not help relieve your symptoms after three months, then you may need further evaluation.
References:
King, William, et al. “Human blood‐based anti‐inflammatory solution inhibits osteoarthritis progression in a meniscal‐tear rat study.” Journal of Orthopaedic Research 35.10 (2017): 2260-2268.
King W, van der Weegen W, Van Drumpt R, Soons H, Toler K, Woodell-May J, “White blood cell concentration correlates with increased concentration of IL-1ra and improvement in WOMAC pain scores in an open-label safety study of autologous protein solution.” Journal of Experimental Orthopaedics. 2016;3:9.
Kon E, Engebretsen L , Peter Verdonk P, Nehrer S and Filardo G. “Clinical Outcomes of Knee Osteoarthritis Treated with an Autologous Protein Solution. A1-year Pilot Double-Blinded Randomized Control Trial. American Journal of Sports Medicine, Oct. 2017.
Kon E, Engebretsen L , Peter Verdonk P, Nehrer S and Filardo G. “Two-year Clinical Outcomes of An Autologous Protein Solution Injection For Knee Osteoarthritis.” ICRS 14th World Congress presented 2018.
Matuska, Andrea, et al. “Autologous solution protects bovine cartilage explants from IL‐1α‐and TNFα‐induced cartilage degradation.” Journal of Orthopaedic Research 31.12 (2013): 1929-1935.
van Drumpt, Rogier AM, et al. “Safety and treatment effectiveness of a single autologous protein solution injection in patients with knee osteoarthritis.” BioResearch open access 5.1 (2016): 261-268.
We are here for you
Request Your Appointment Today!

WELCOME TO RRMG!
Welcome to a Family RRMG Healthcare
Rogers Regenerative Medical Group (RRMG) provides world-class Regenerative Orthopaedics and Sports Medicine specializing in the comprehensive treatment of the musculoskeletal system and the use of specific treatment types best suited for sports injuries and a variety of other health issues. The treatments range from non-operative, minimally invasive, regenerative therapies or a combination.
Other services include metabolic medicine, exercise physiological testing, weight management, biomechanical analysis, ergonomic assessments, gait and postural analysis to identify and measure abnormal movement patterns and muscle imbalances, running medicine, nutritional guidance and weight management.

+44 (0) 20 7112 5400
Contact us for a
Consultation!
